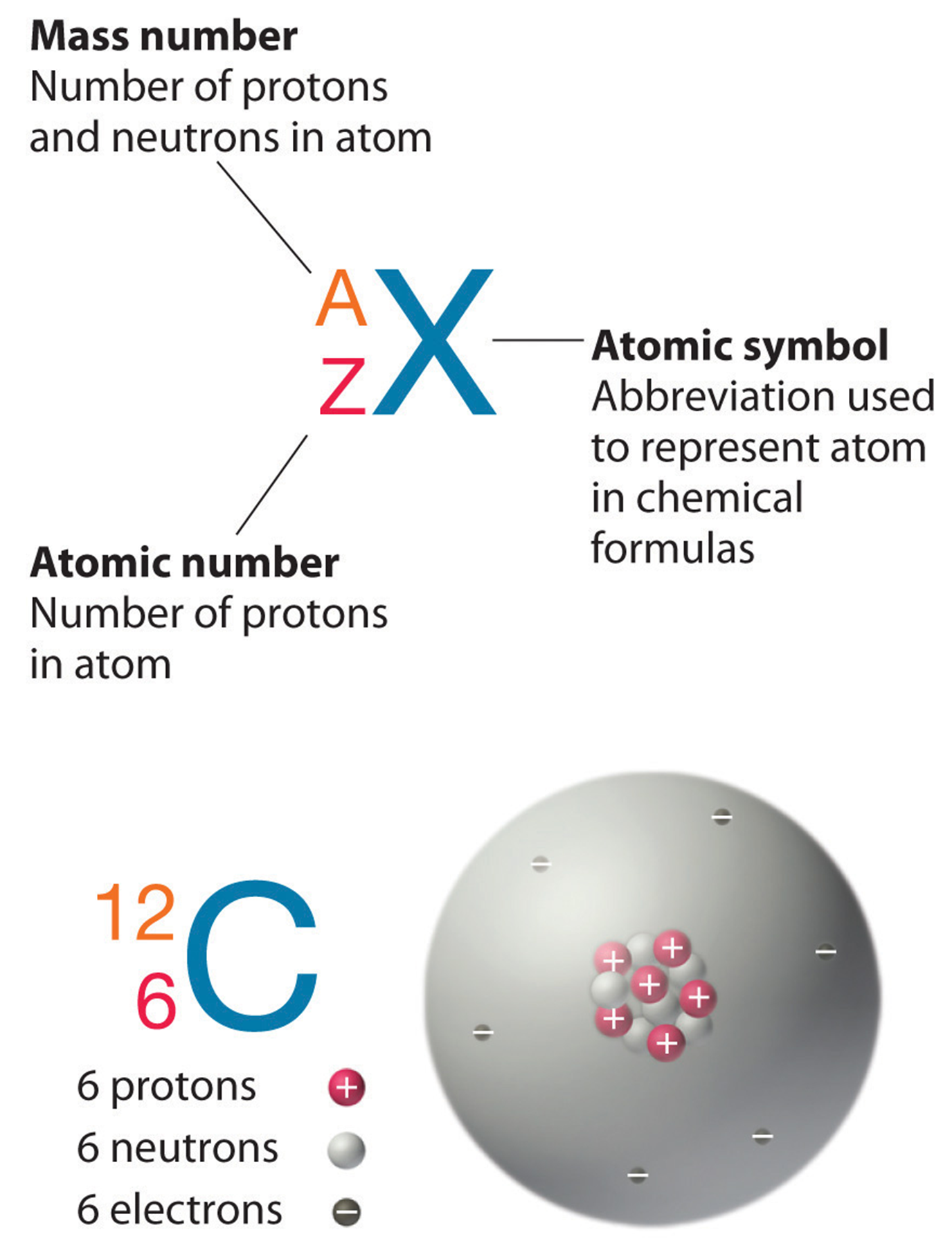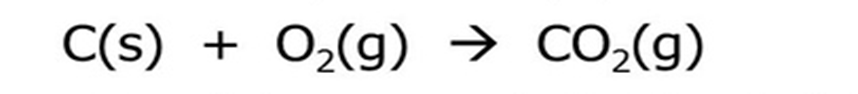Module 2: Science, Matter, Energy, and Systems
Module 2: Science, Matter, Energy, and Systems
How scientists study the natural world
Evidence, hypotheses, theories, and laws
Matter, energy, and their transformations
Systems thinking and feedbacks in nature
Links to sustainability and environmental change
Learning Objectives
By the end of this module, you should be able to:
Describe how scientific knowledge is developed and evaluated
Distinguish hypotheses, theories, and scientific laws
Explain basic properties of matter and energy
Apply the laws of conservation and thermodynamics
Describe systems, feedback loops, and tipping points
Connect physical and biological principles to environmental issues
How Do Scientists Learn About Nature?
- Explain how the natural world works
- Focus on cause-and-effect relationships
- Rely on observation, evidence, and testing
- Scientific knowledge develops over time
Science Starts with Questions
Using Experiments to Test Ideas
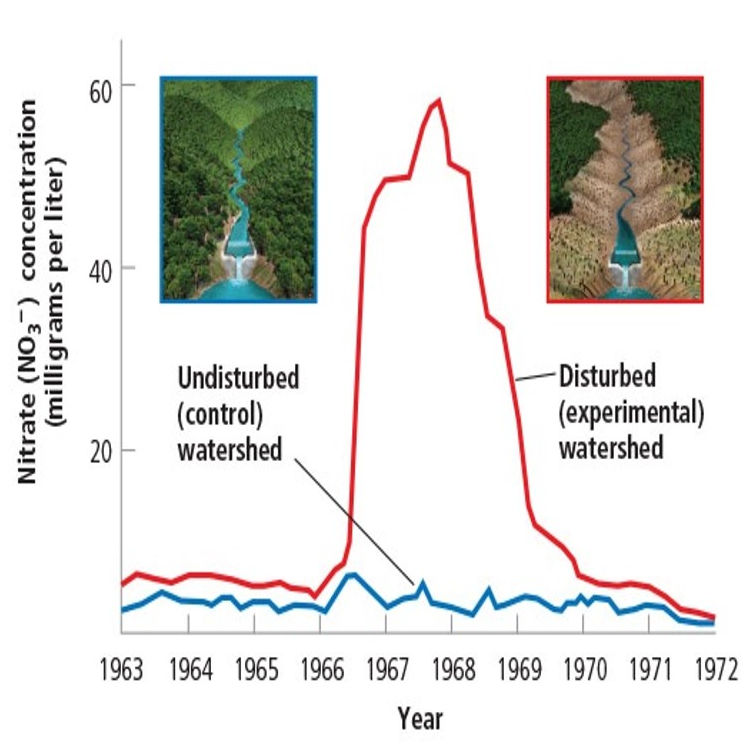
Example: Forests and Water Loss
What the Experiment Showed
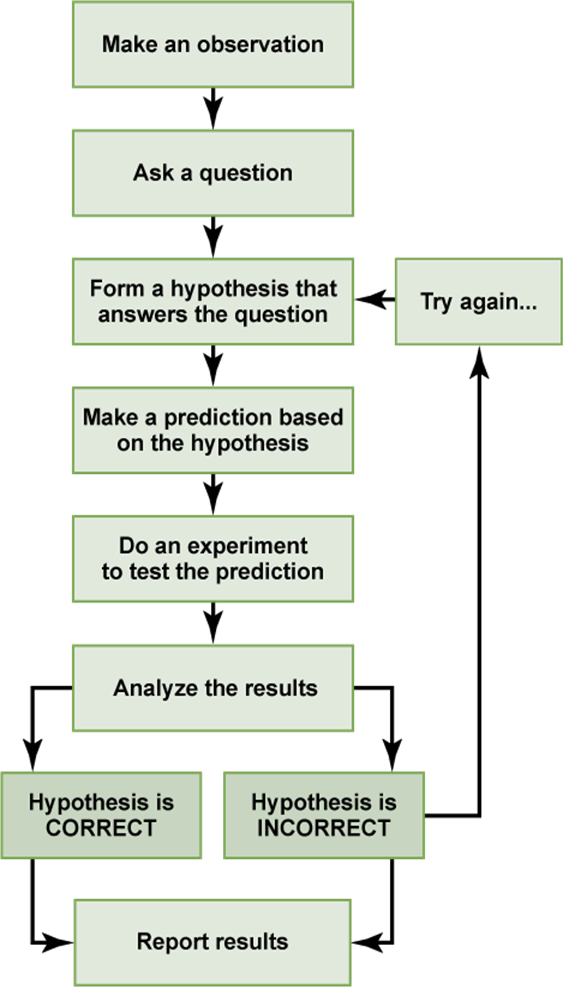
What Do Scientists Do?
The Scientific Process (Simplified)
Hypotheses
Scientific Theories
Scientific Laws
- A description of a consistent pattern in nature
- Often expressed mathematically
- Describes what happens, not why it happens
- Highly reliable and repeatable
- Revised only with new evidence
- Laws are not “proven theories”
- Theories do not graduate into laws
- They answer different questions:
- Laws: What happens?
- Theories: Why and how does it happen?
Hypotheses vs. Theories vs. Laws
- Hypothesis: tentative, testable idea
- Theory: well-supported explanation
- Law: consistent description of behavior
- Different roles, not levels of certainty
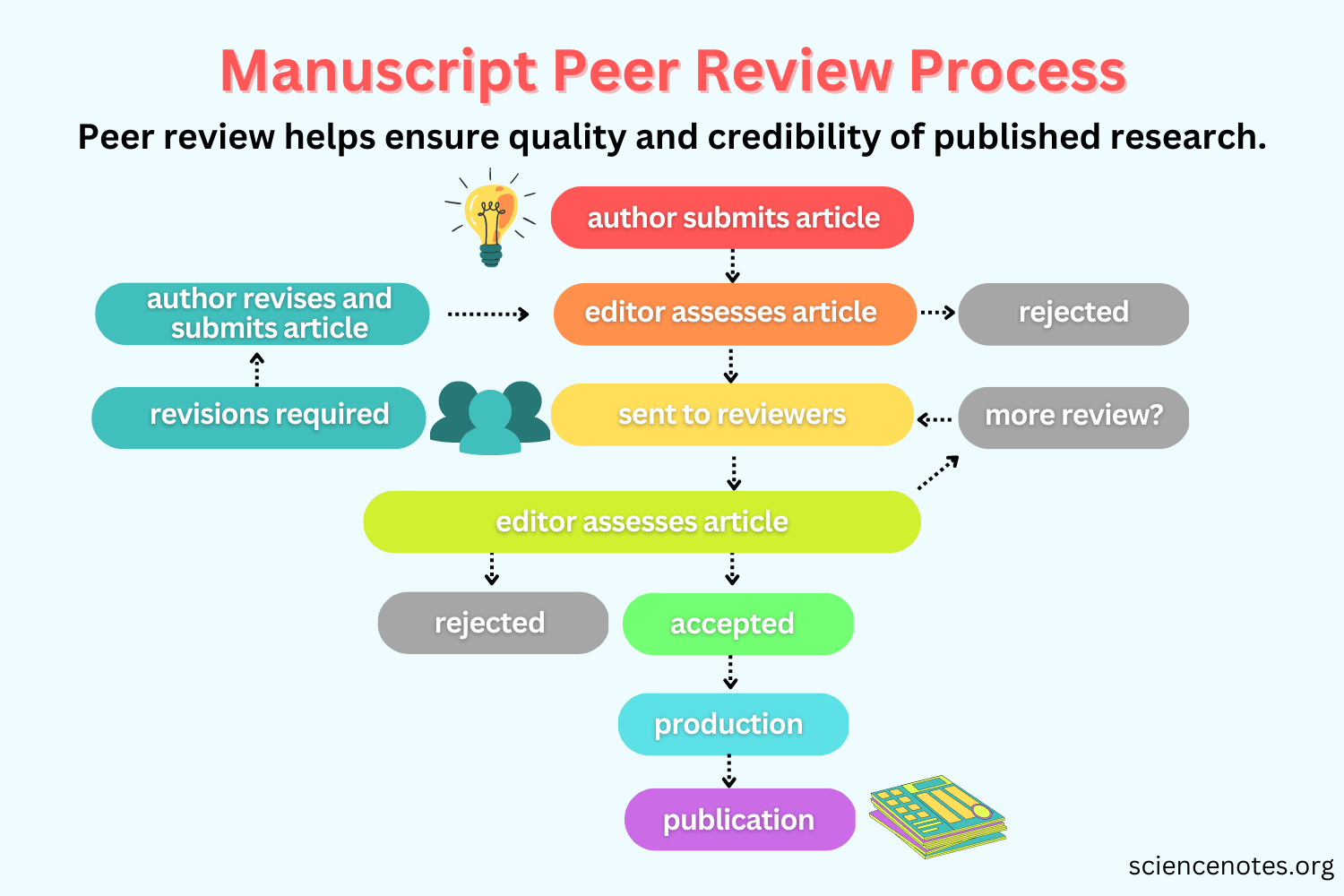
Evaluating Scientific Knowledge
Peer review
Independent evaluation by other scientists in the fieldReplication of results
Findings can be reproduced using the same methodsTransparent methods
Clear description of data, methods, and analysesStrength of evidence
Multiple lines of evidence support the same conclusion
Limits of Science
No absolute proof
Scientific conclusions are always open to revisionMeasurement uncertainty
Observations and measurements contain inherent errorPotential for bias
Scientists are human and can be influenced by assumptionsPeer review reduces bias
Independent review helps identify errors and bias
What Is Matter and What Happens When It Undergoes Change?
Matter Consists of Elements and Compounds
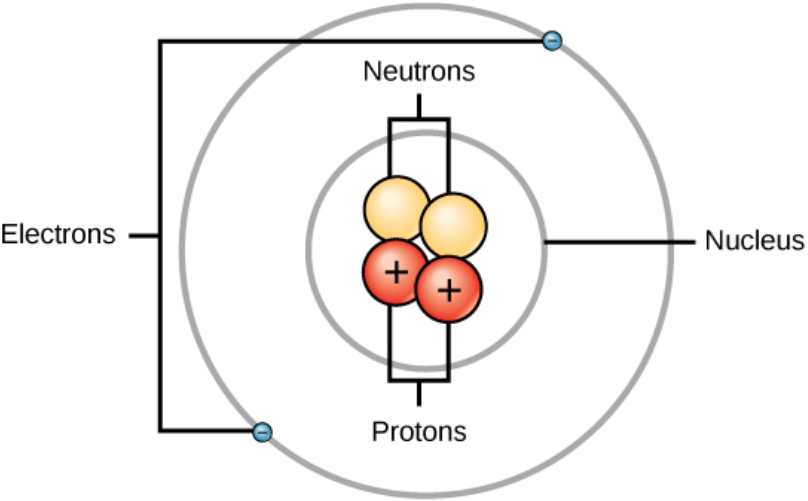
An Atom Is the Basic Building Block of Matter
Atomic Number, Atomic Mass, and Element
Atoms differ in numbers of subatomic particles
Atomic number determines the element
- Number of protons in the atomic nucleus
Mass number
- Total number of protons and neutrons in the nucleus of an element’s atoms
Element
- A pure substance that consists only of atoms with the same number of protons
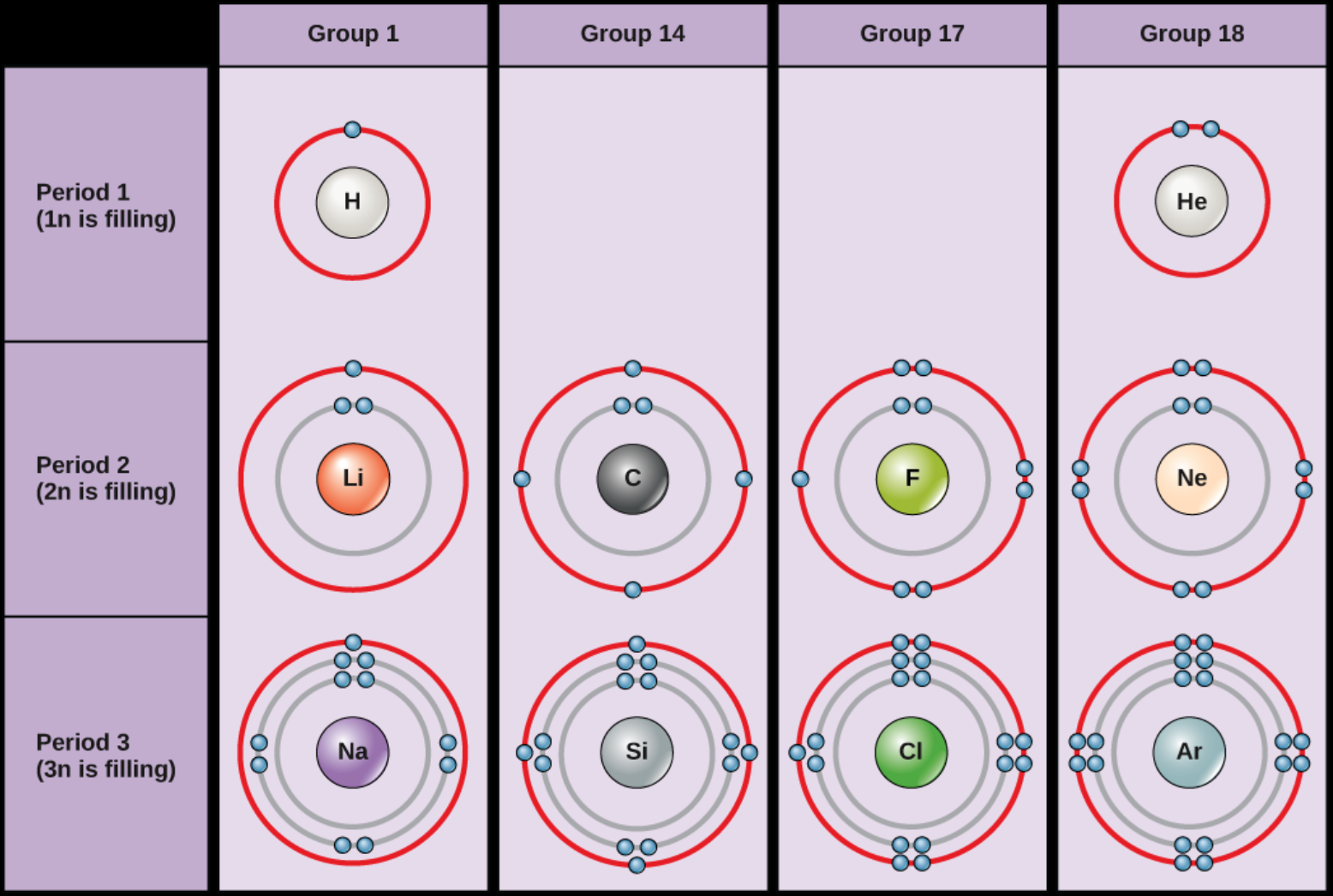
Why Electrons Matter (shell model)
- Electrons occupy energy levels (shells) around the nucleus
- Outer-shell electrons determine how atoms interact
- Atoms with incomplete outer shells are chemically reactive
- Atoms gain, lose, or share electrons to become more stable
- Chemical reactivity controls nutrient availability, toxicity, and pollutant behavior in the environment
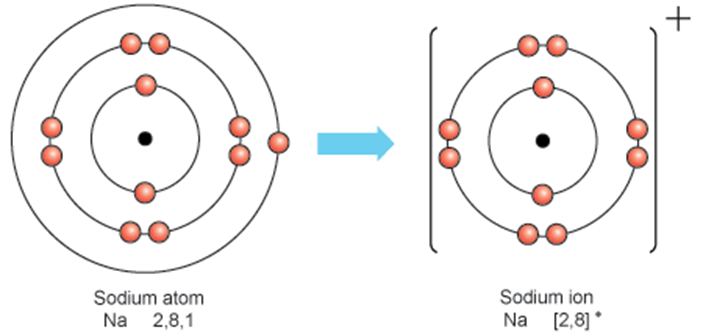
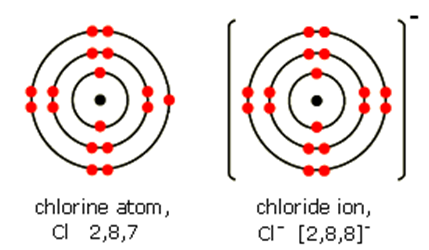
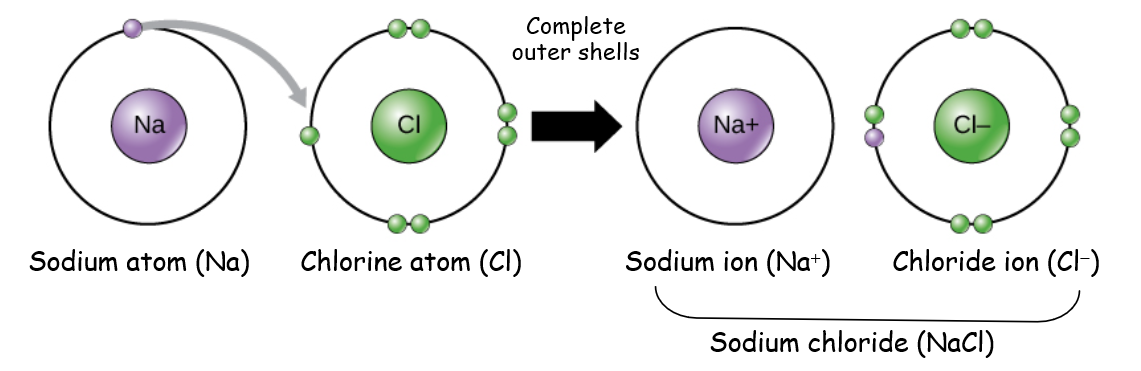
Ions
Atoms with a net electric charge
Formed when atoms gain or lose electrons
Loss of electrons → positively charged ion (cation)
Gain of electrons → negatively charged ion (anion)
Ionic form affects solubility, mobility, and biological availability in the environment
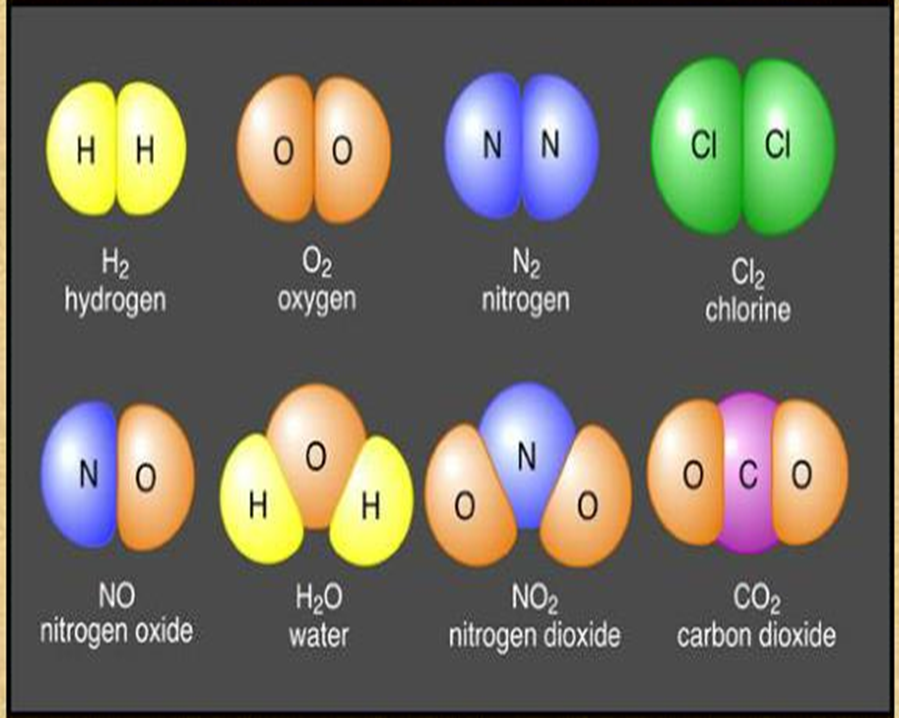
Ionic Bonds
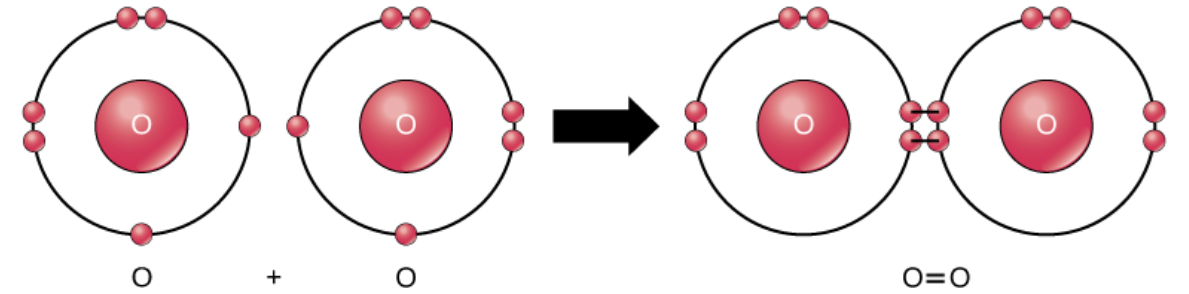
Covalent Bonds
Sharing of electrons between atoms to form covalent bonds.
Stronger and much more common than ionic bonds in the molecules of living organisms.
Commonly found in carbon-based organic molecules, such as our DNA and proteins.
Also in inorganic molecules like H2O, CO2, and O2.
One, two, or three pairs of electrons may be shared, making single, double, and triple bonds, respectively. More bonds = stronger; triple bonds are the strongest
So, why care about ions and compounds?
- So, understanding the chemistry of ions and compounds allows us to measure them.
Remember the trees in the valley?
Scientists learned how to measure nitrate and nitrite ions in water to determine the concentration of these ions
- Reduce nitrate with zinc and react with naphthylethylenediamine under acid conditions to produce a red compound - 550mn.
Determined that lack of trees washed out nitrates from the soil, increasing in concentration in the water runoff.
Acids and Bases
Acid
A chemical compound that donates H+ ions to solutions.
Aqueous solutions with a pH less than 7 are said to be acidic
Base
A compound that accepts H+ ions and removes them from solution.
Aqueous solutions with a pH greater than 7 are basic or alkaline.
Pure water has a pH very close to 7
If an equal number of these ions are present in a solution the pH will not change as it is said to be buffered.
A pH Scale
Organic compounds are the chemicals of life
Organic compounds have at least two carbon atoms and various other elements
Hydrocarbons: contain carbon and hydrogen atoms
Simple carbohydrates: contain carbon, hydrogen, and oxygen
Polymers: simple organic compounds (monomers) chemically bonded together
Polymers are essential to life
What are the major types of polymers?
Complex carbohydrates: two or more monomers of simple sugars such as glucose
Proteins: formed by amino acids (that are monomers)
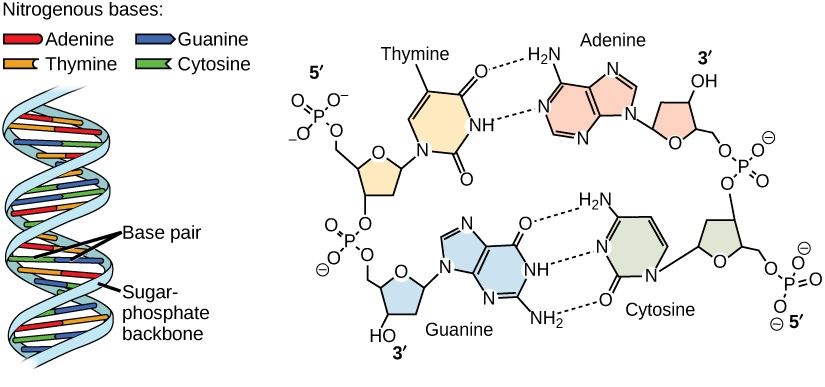
Nucleic acids: such as DNA and RNA that are formed by nucleotides (that are monomers)
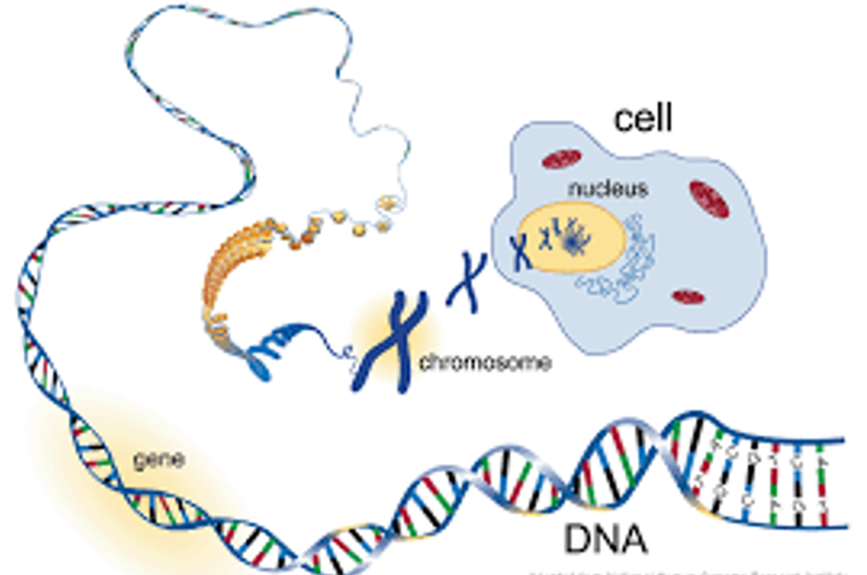
The structure of DNA: The best known biomolecule
Matter Comes to Life Through Cells, Genes, and Chromosomes
Matter undergoes physical, chemical, and nuclear changes
Physical changes
Change form or state (e.g., ice to water) without changing chemical identityChemical changes
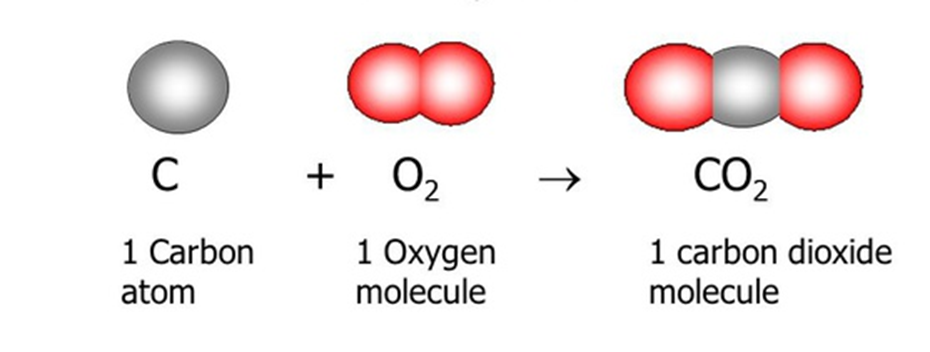
Rearrangement of atoms to form new substancesChemical equations
Represent reactants, products, and conservation of atoms
Cannot create or destroy atoms
Law of conservation of matter
Matter is neither created nor destroyed in physical or chemical changesAtoms are rearranged, not created or lost
Balanced chemical equations
Ensure the same number of each type of atom on both sides of a reaction
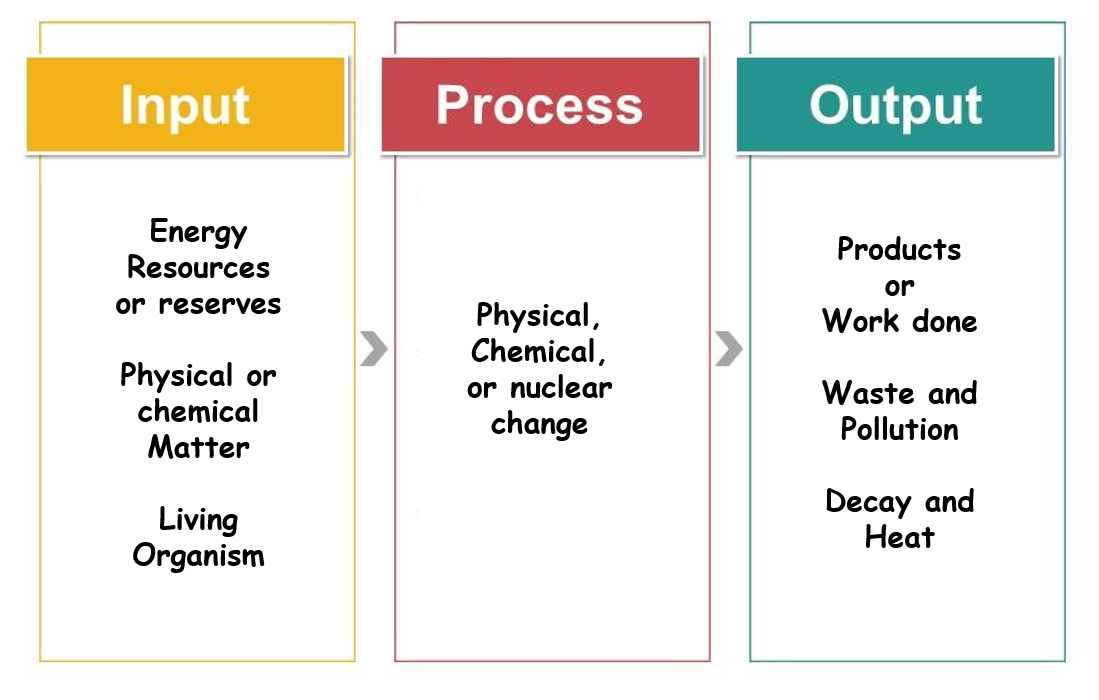
What Is Energy and What Happens When It Undergoes Change?
Energy
The ability to do work or transfer heatFirst law of thermodynamics
Energy is conserved; it changes form but is not created or destroyedSecond law of thermodynamics
Energy transformations result in lower-quality, less usable energy
Energy
Energy Comes in Many Forms: Kinetic Energy
Energy comes in many forms: potential energy
Potential energy
Stored energy that has the capacity to do workExample:
- Water stored behind a dam
Potential energy can be converted to kinetic energy
- Flowing water spins turbines
- Turbines generate electrical energy
Some Types of Energy Are More Useful Than Others
- High-quality energy
Concentrated energy with high capacity to do work- Examples: high-temperature heat, concentrated sunlight, strong wind
- Low-quality energy
Dispersed energy with limited ability to do work- Example: heat lost to the environment
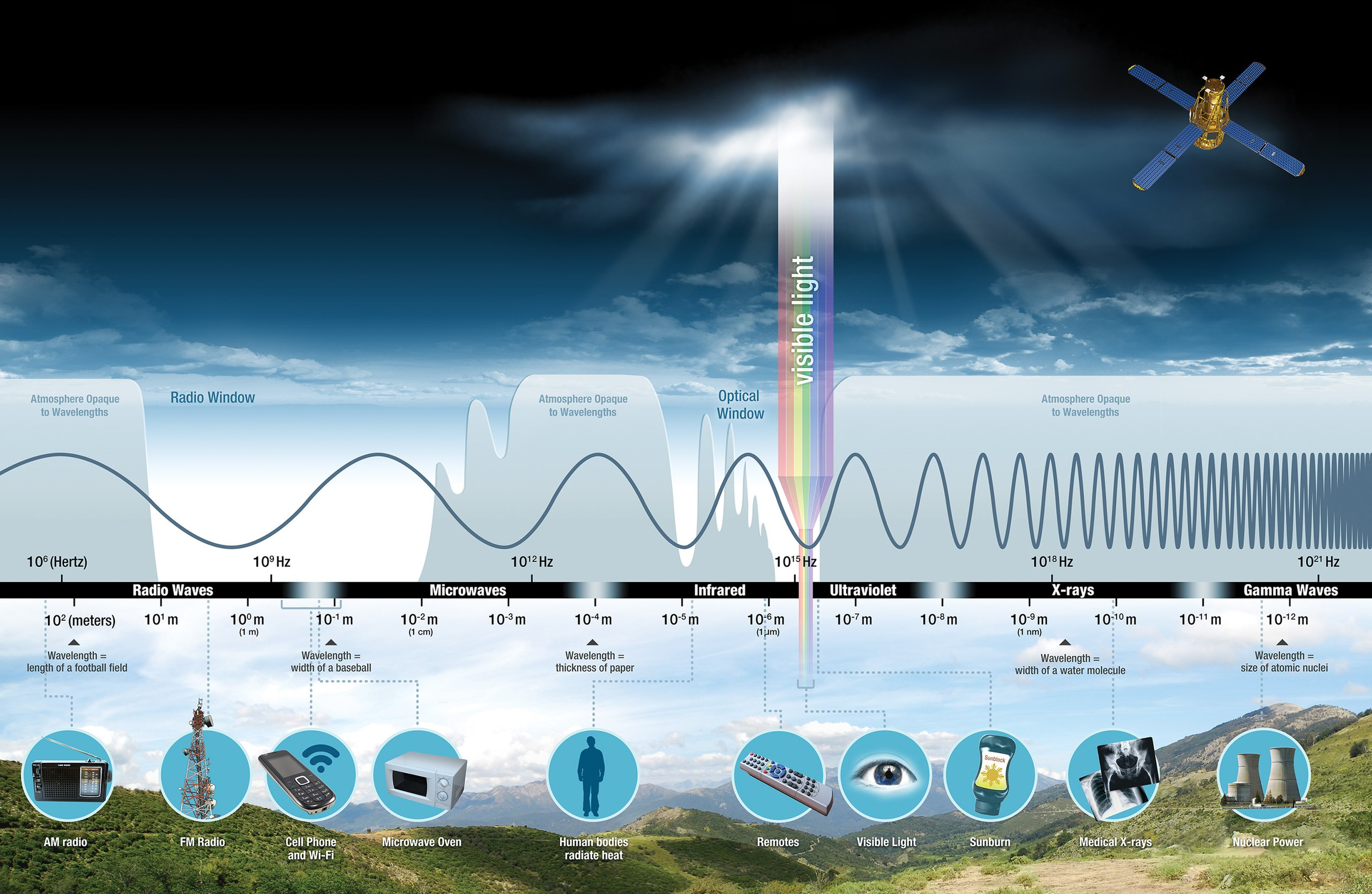
Solar Energy and Earth Systems
- Solar energy arrives as electromagnetic radiation
- Spans a range of wavelengths (the electromagnetic spectrum)
- Primary energy source for:
- Climate and weather
- Photosynthesis
- Ecosystems and food webs
- Drives most natural processes on Earth
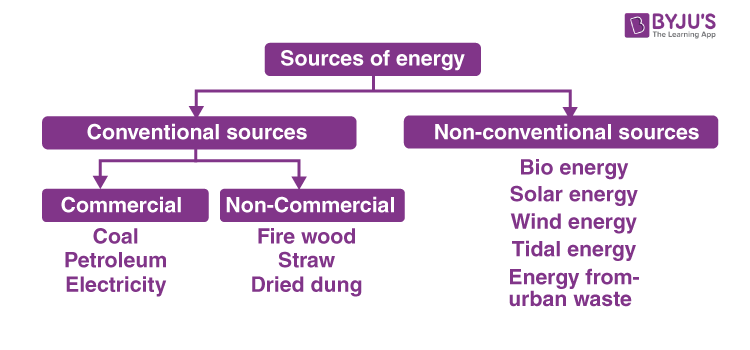
Human Energy Use: Commercial Energy
Commercial energy
Energy extracted, processed, and sold by humansIncludes:
- Fossil fuels (coal, oil, natural gas)
- Electricity
- Nuclear and renewable sources
Most commercial energy currently comes from fossil fuels
- Nonrenewable
- Major environmental and climate impacts
What Are Systems and How Do They Respond to Change?
Living and Nonliving Systems
- Nonliving systems
- Do not grow or adapt to environmental change
- Function remains largely fixed
- Do not grow or adapt to environmental change
- Living systems
- Respond to environmental change
- Adjust size, structure, and behavior
- Respond to environmental change
- Systems become unsustainable when:
- Inputs are used faster than they are replenished
- Outputs exceed the environment’s capacity to absorb or dilute them
- Inputs are used faster than they are replenished
Ecological System Feedback Loops
Feedback loop
Occurs when a system output feeds back as an input, influencing future system behaviorPositive feedback
Amplifies change, pushing the system further in the same directionNegative feedback
Dampens change, promoting stabilityExample of positive feedback:
- Vegetation loss increases erosion and nutrient loss
- Poor soil conditions cause further vegetation decline
- System becomes increasingly degraded over time
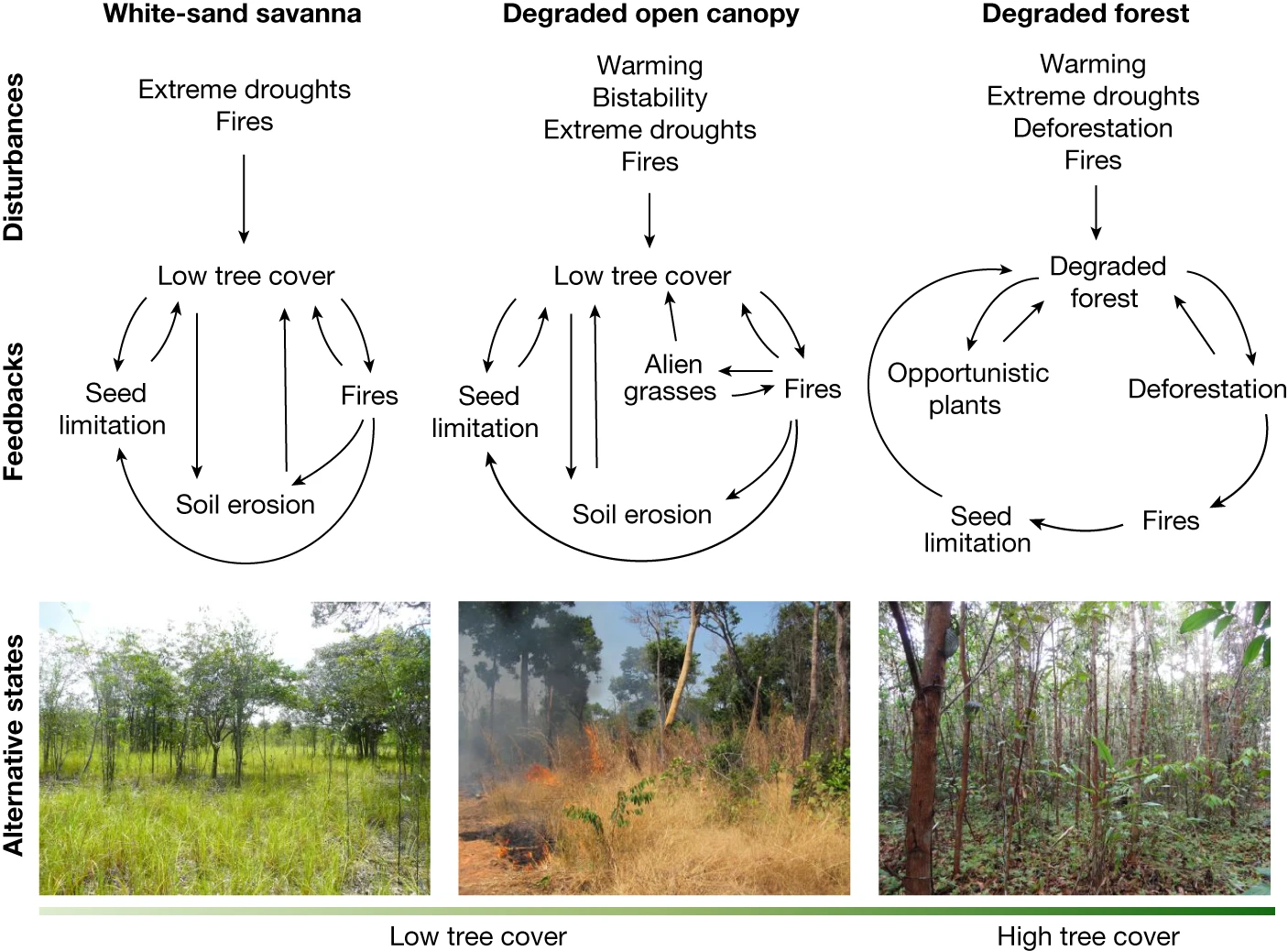
Ecological Tipping Points
Tipping point
A threshold beyond which a system shifts rapidly to a new stateOften triggered by sustained pressure or positive feedbacks
After a tipping point:
- Change can be difficult or impossible to reverse
- System structure and function may fundamentally change
Examples:
- Forest to grassland transitions
- Coral reef collapse
Negative (Corrective) Feedback Loops
Negative feedback loop
Counteracts change and promotes system stabilityExample: thermostat
- Measures temperature
- Turns heating or cooling on or off to maintain a set point
- Measures temperature
Environmental example: aluminum recycling
- Used cans become inputs rather than waste
- Reduces mining and manufacturing demand
- Lowers energy use, pollution, and material extraction
- Used cans become inputs rather than waste

BIOL 346 An Ecological Perspective | Spring 2026